The integration of generative artificial intelligence into the medical field has reached a significant milestone with the development of BUSGen, a foundation model that is currently redefining the precision of breast cancer diagnostics. While traditional AI systems in healthcare have historically been designed for narrow, singular tasks, this new architecture leverages a massive dataset of 3.5 million images to establish a versatile baseline for clinical analysis. This shift toward foundational models allows for a more holistic understanding of medical imagery, moving beyond simple pattern recognition toward a deep synthesis of pathological characteristics. By outperforming human experts in diagnostic accuracy, the system addresses critical gaps in early detection and provides a scalable solution for the global healthcare community. The arrival of such technology suggests that the bottleneck of human subjectivity in ultrasound interpretation is finally being overcome through large-scale data training and advanced generative techniques.
Specialized AI Architecture and Ultrasound Complexity
Addressing the Nuances of Breast Imaging
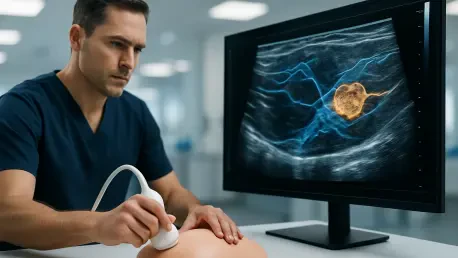
Breast ultrasound has long been regarded as one of the most challenging modalities in medical imaging due to its inherent “operator-dependent” nature and the presence of significant acoustic noise. Unlike a standard CT scan or MRI, which produces relatively uniform results, an ultrasound image changes drastically depending on the angle of the transducer, the pressure applied by the technician, and the specific settings of the machine used. These variables often lead to artifacts that can mimic or hide actual lesions, creating a high cognitive load for radiologists who must distinguish between benign shadows and malignant masses. BUSGen navigates this complexity by internalizing the “latent space” of these images, meaning it has learned to recognize the underlying structures of breast tissue regardless of the external noise or the quality of the capture. This fundamental understanding allows the model to act as a robust filter, identifying clinical signals that might be obscured by the grainy texture typical of ultrasound scans.
Beyond merely identifying anomalies, the foundation model architecture enables the system to adapt to various downstream clinical tasks without the need for exhaustive retraining. In the current landscape of 2026, where medical facilities utilize a diverse range of hardware from different manufacturers, the ability of an AI to remain consistent across different imaging environments is invaluable. The model’s training on millions of diverse images ensures that it understands the “grammar” of human anatomy, recognizing how healthy fibrous, fatty, and glandular tissues interact. This deep-seated knowledge provides a level of reliability that matches or exceeds the diagnostic consistency of veteran radiologists, who often rely on years of subjective experience to navigate the same visual ambiguities. By standardizing the interpretation of these complex images, the technology provides a stable foundation for more accurate screenings, potentially reducing the number of unnecessary biopsies while ensuring that actual malignancies are caught earlier.
Navigating Technical Variability in Diagnostic Workflows
The challenge of technical variability extends to the specific characteristics of different patient populations, where breast density and tissue composition can vary significantly based on age, genetics, and hormonal factors. Conventional AI models often struggle when presented with data that deviates from their narrow training set, leading to “model drift” or a loss of accuracy in real-world clinical settings. BUSGen avoids this pitfall by utilizing its broad foundational knowledge to maintain performance across diverse demographic groups. Because it was trained on such a vast and varied dataset, it has encountered nearly every conceivable variation in breast anatomy, allowing it to interpret scans with a level of nuance previously thought to be exclusive to human experts. This adaptability is particularly crucial in multi-center clinical trials where data comes from various sources with differing protocols.
Furthermore, the implementation of this foundation model into existing diagnostic workflows simplifies the role of the medical technologist. In many instances, the AI can provide real-time feedback during the scanning process, suggesting a change in probe angle or identifying areas that require a more detailed look. This collaborative dynamic between the machine and the operator reduces the likelihood of “non-diagnostic” images that require a patient to return for a follow-up appointment. By mitigating the effects of human error at the point of acquisition, the system ensures that the data being analyzed is of the highest possible quality. This proactive approach to imaging not only streamlines the operations of a busy radiology department but also significantly enhances the patient experience by providing faster, more reliable results during the initial visit.
The Power of Synthetic Data and Adaptation
Overcoming Data Scarcity and Privacy Barriers
One of the primary obstacles to advancing medical AI has been the difficulty of accessing high-quality, annotated datasets due to stringent privacy regulations such as HIPAA and GDPR. Researchers often find themselves in a catch-22 where they need millions of images to train a reliable model, yet they cannot share patient data across institutional lines without risking legal repercussions or ethical breaches. BUSGen solves this dilemma through its ability to generate high-fidelity synthetic data. These are not merely “fake” images; they are mathematically accurate reconstructions that reflect the statistical and pathological properties of real-world cancer cases. Because these images do not belong to any actual patient, they can be shared freely between research institutions, allowing for a level of global collaboration that was previously impossible. This creates a “data engine” that can fuel the development of other specialized medical tools without ever compromising patient confidentiality.
The impact of synthetic data is most profound when addressing rare forms of breast cancer or specific pathological subtypes that are infrequently documented in traditional databases. In a typical clinical setting, an AI might only see a few dozen examples of a rare malignancy, which is insufficient for it to learn how to identify that disease reliably. However, by using its generative capabilities, BUSGen can create thousands of variations of these rare cases, effectively “teaching” itself and other models how to recognize the subtle markers of uncommon diseases. This methodology also helps to correct class imbalances in training sets, where healthy “normal” scans usually far outnumber “pathological” ones. By intentionally generating more examples of disease, researchers can ensure that the AI is just as proficient at finding cancer as it is at confirming health. This balanced training leads to a much more sensitive and specific diagnostic tool that is prepared for the widest possible range of clinical scenarios.
Few-Shot Learning and Rapid Clinical Deployment
In the rapidly evolving field of medical technology, the ability to quickly adapt to new hardware or emerging diseases is a critical competitive advantage. Standard AI models typically require thousands of new, labeled images to learn a new task, a process that can take months of data collection and expert annotation. In contrast, BUSGen utilizes a “few-shot adaptation” technique, which allows it to master a new specific pathology or adjust to a new type of ultrasound machine using only a handful of examples. This is possible because the model already possesses a comprehensive understanding of the fundamentals of ultrasound imaging; it only needs to learn the specific “delta” or difference presented by the new task. This capability drastically reduces the time and cost associated with deploying AI updates in a clinical environment, ensuring that the latest diagnostic breakthroughs can reach patients in weeks rather than years.
This agility is particularly relevant as the industry moves toward more specialized and localized medical care. For example, a hospital in a specific geographic region might encounter a higher prevalence of certain tissue types or specific health conditions. With few-shot learning, the facility can fine-tune the foundation model to its specific local population using a very small set of their own data. This ensures that the AI remains highly accurate for the people it serves, rather than relying on a generalized average that might not be perfectly applicable. Moreover, as new ultrasound technologies, such as 3D automated breast ultrasound (ABUS), become more common, the foundation model can be adapted to these new formats with minimal friction. This future-proofs the investment in AI, as the core intelligence of the system remains relevant even as the peripheral hardware continues to evolve throughout 2026 and beyond.
Clinical Performance and Diagnostic Benchmarks
Surpassing Human Experts in Detection Sensitivity
The validation of BUSGen involved a rigorous head-to-head comparison with nine board-certified radiologists who specialize exclusively in breast cancer detection. In these trials, the model demonstrated a staggering 16.5% improvement in sensitivity over the human experts, a margin that is considered highly significant in the medical community. Sensitivity refers to the ability of a test to correctly identify those with the disease, and in the context of breast cancer, every percentage point represents lives saved through earlier intervention. Human experts, despite their extensive training, are susceptible to “observer fatigue” and cognitive biases, especially during long shifts where they must review hundreds of similar-looking scans. The AI, however, maintains a consistent level of scrutiny for every single frame, ensuring that subtle micro-calcifications or architectural distortions are never overlooked.
This leap in performance is not just a matter of processing power but a result of the model’s ability to see patterns that are virtually invisible to the human eye. Radiologists are trained to look for specific, documented signs of malignancy, such as irregular borders or posterior shadowing. While BUSGen certainly looks for these same signs, it also analyzes millions of pixel-level correlations that define the “texture” of the tissue. This allows the model to flag suspicious areas that a human might dismiss as normal glandular variation. By serving as a persistent, high-accuracy second opinion, the system effectively acts as a safety net. When a radiologist and the AI both agree on a diagnosis, the confidence level of the clinical team increases. When they disagree, it prompts a more thorough investigation, such as a targeted biopsy or a follow-up MRI, which can lead to the discovery of early-stage cancers that would have otherwise gone undetected until a later, more dangerous stage.
Reducing Missed Diagnoses and Clinical Errors
The reduction of “false negatives”—cases where cancer is present but not detected—is perhaps the most vital contribution of this technology to modern oncology. A missed diagnosis in breast cancer often means that by the time the disease is finally caught, it has progressed to a point where the treatment must be far more aggressive, involving systemic chemotherapy or radical surgery. By increasing detection sensitivity by over 16%, BUSGen significantly lowers the threshold for early discovery, which is directly correlated with higher survival rates. The model’s consistency ensures that the quality of a patient’s screening does not depend on which doctor happens to be on call or how many hours they have been working. This standardization of care is a major step toward eliminating the “postcode lottery” in healthcare, where the accuracy of a diagnosis can vary based on the specific expertise available at a local clinic.
Furthermore, the system helps to address the issue of “false positives,” which cause unnecessary anxiety and lead to invasive procedures that carry their own risks. Because the model has such a deep understanding of the “latent space” of healthy tissue, it is better equipped to distinguish between benign cysts and malignant solid masses. This precision allows radiologists to be more selective about which cases they refer for biopsy. In a high-volume screening environment, reducing the number of unnecessary follow-ups not only saves money for the healthcare system but also alleviates the significant emotional and physical toll on patients. The transition from subjective human interpretation to a data-driven, AI-augmented workflow represents a fundamental shift in how diagnostic medicine is practiced, prioritizing objective evidence and statistical probability over clinical “gut feeling.”
Prognostic Insights and Personalized Medicine
Predicting Disease Progression and Treatment Paths
While the primary role of ultrasound has traditionally been diagnostic, BUSGen is expanding the modality’s utility into the realm of prognosis. By identifying specific imaging biomarkers—microscopic patterns in the tissue architecture that indicate the biological behavior of a tumor—the model can offer insights into how a disease is likely to evolve over time. For instance, the AI can analyze the “vascularity” and “stiffness” of a lesion through the ultrasound data to predict whether a tumor is likely to be aggressive and fast-growing or indolent and slow-moving. This prognostic capability is a cornerstone of personalized medicine, moving away from a one-size-fits-all approach and toward treatments that are tailored to the specific genetic and structural profile of an individual’s cancer.
The ability to predict patient outcomes directly from an ultrasound image is a revolutionary development that could bypass the need for some expensive genomic testing. If the model identifies features associated with a high risk of metastasis, the clinical team can proactively opt for more intensive systemic therapies, such as neoadjuvant chemotherapy, before the cancer has a chance to spread. Conversely, for tumors that the model identifies as low-risk, physicians might recommend a “de-escalation” of treatment, sparing the patient from the debilitating side effects of radiation or chemotherapy. This level of insight allows for a much more nuanced management of the disease, where the intensity of the medical intervention is perfectly calibrated to the actual threat posed by the tumor. As we move through 2026, the integration of these prognostic AI tools into daily practice is helping to define a new era of “precision oncology” that is both more effective and more humane.
Enhancing Long-Term Patient Management
Beyond the initial treatment phase, the prognostic insights provided by BUSGen play a critical role in long-term survivorship and monitoring. For patients who have completed their primary treatment, the model can be used to analyze follow-up scans with a high degree of sensitivity to the earliest signs of recurrence. Because the system can compare current images with historical data at a pixel-by-pixel level, it is exceptionally good at detecting subtle changes in tissue that might indicate the cancer is returning. This longitudinal analysis provides a level of surveillance that is far more detailed than what can be achieved through manual comparison of printouts or digital files by a human radiologist. By catching recurrences at their most microscopic stage, the AI ensures that salvage therapies can be initiated immediately, significantly improving the chances of long-term survival.
Moreover, these insights contribute to a more comprehensive understanding of how different patients respond to specific therapies. By aggregating the prognostic data from thousands of cases, researchers can begin to identify which imaging signatures are associated with positive responses to new immunotherapies or targeted drugs. This creates a feedback loop where the AI not only helps the individual patient but also contributes to the broader scientific knowledge base. In this way, the foundation model acts as a bridge between the clinic and the laboratory, turning every ultrasound scan into a valuable data point for medical research. This shift toward a data-centric view of patient management ensures that every clinical decision is backed by a global repository of knowledge, ultimately leading to better health outcomes and a more efficient use of medical resources.
Global Health Impact and Technical Scaling
Democratizing Access and Future Frameworks
The global shortage of trained radiologists is one of the most significant barriers to effective cancer screening, particularly in low-to-middle-income countries where access to specialized medical training is limited. BUSGen offers a scalable solution to this crisis by acting as an automated “expert-in-a-box.” In regions where there are not enough doctors to read every ultrasound, the AI can perform the initial screening, filtering out the vast majority of normal cases and flagging only the most suspicious ones for review by a remote specialist. This “triage” system ensures that the limited time of human experts is focused on the patients who need it most, dramatically increasing the capacity of the healthcare system. By lowering the expertise barrier required to perform high-quality screenings, the technology helps to democratize access to life-saving diagnostics.
Technically, the success of BUSGen serves as a blueprint for the future of AI across all medical disciplines. The researchers’ demonstration of the “scaling effect”—where more synthetic data directly leads to higher performance—proves that the foundation model approach is the most viable path forward for medical AI. This methodology is already being adapted for other fields, such as cardiology for echocardiograms and neurology for brain scans. By establishing a central “brain” that understands the fundamentals of medical imaging, the industry can avoid the inefficiencies of building thousands of small, disconnected models. This unified framework allows for a more rapid cross-pollination of ideas and technical breakthroughs, accelerating the pace of innovation across the entire healthcare sector. As these systems become more integrated, we can expect to see a more interconnected and intelligent global health infrastructure that leaves no patient behind.
Actionable Strategies for Implementation
To fully realize the benefits of foundation models like BUSGen, healthcare organizations must move toward a strategy of “AI integration” rather than mere “AI adoption.” This involves not only installing the software but also redesigning clinical workflows to maximize the synergy between human intuition and machine precision. Facilities should prioritize the development of robust data pipelines that allow for the secure use of synthetic data to fine-tune models for local demographics. Furthermore, there must be an industry-wide commitment to transparency and validation; while the model’s sensitivity is high, human oversight remains essential to interpret the findings within the broader clinical context of the patient’s history and symptoms. Educational programs for radiologists should be updated to include “AI literacy,” teaching professionals how to effectively interact with and challenge the outputs of these advanced systems.
Looking forward, the focus must shift toward the creation of multi-modal foundation models that can synthesize data from ultrasound, mammography, MRI, and even genetic profiles into a single diagnostic picture. The success of BUSGen in the ultrasound niche is a clear indicator that the technology is ready for this next level of complexity. By adopting a proactive stance on AI-augmented diagnostics, the medical community can ensure that it is prepared for the challenges of an aging global population and the increasing demand for precision care. The ultimate takeaway from the emergence of this technology is that the combination of massive datasets and generative architectures has finally provided a tool that can truly match the complexity of human biology, offering a pathway to a future where breast cancer is caught earlier, treated more effectively, and managed with unprecedented accuracy.