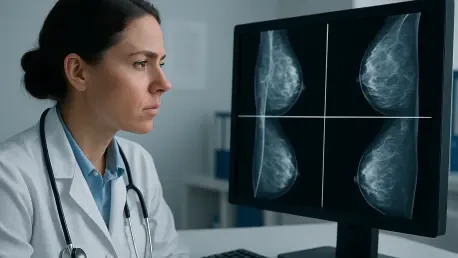
The global medical community is currently navigating a profound period of transformation as the integration of advanced machine learning into clinical workflows fundamentally redefines traditional standards of care. At the heart of this evolution lies a mounting tension between the rapid acceleration of high-fidelity clinical evidence and the static regulatory frameworks maintained by the Food and Drug Administration. This conflict has been brought into sharp focus by the recent publication of updated data from the Mammography Screening with Artificial Intelligence (MASAI) trial in The Lancet. As a randomized, longitudinal study conducted in Sweden, the MASAI trial provides what many specialists consider the most rigorous evidence to date regarding the efficacy of AI-augmented diagnostics. By comparing the established “double reading” protocol against a modernized workflow where a single radiologist works in tandem with an algorithm, the study exposes a significant gap between what technology can achieve and what current regulations permit.
Analyzing the MASAI Trial and the Human-Algorithm Dyad
Shifting Clinical Standards and the Workforce Crisis
The primary findings of the MASAI trial indicate that AI-augmented screening is capable of identifying significantly more cancers while simultaneously reducing the exhaustive workload that typically falls on medical professionals. This revelation is particularly critical given that the United States is currently grappling with a severe and worsening shortage of breast imaging specialists, a crisis that disproportionately affects rural and underserved populations. In many regions, the demand for timely screenings has far outpaced the availability of qualified radiologists, leading to dangerous backlogs and delayed diagnoses. The MASAI data suggests that AI does not simply act as a peripheral enhancement but serves as a vital stabilization mechanism for a fracturing healthcare infrastructure. By demonstrating that a single human reader paired with high-performance software can outperform the traditional two-human model, the study offers a practical solution to the personnel deficit without sacrificing diagnostic integrity.
Beyond the immediate relief of administrative burdens, the integration of AI into the screening process represents a fundamental shift in how diagnostic accuracy is measured and achieved. The MASAI trial proved that the algorithm’s ability to flag subtle anomalies allows radiologists to focus their attention on the most suspicious cases, effectively filtering out noise that often leads to burnout or oversight. This improved efficiency is not merely about speed; it is about the qualitative enhancement of the diagnostic environment. When a healthcare system can maintain or even exceed its baseline performance with half the human resources traditionally required for interpretation, the economic and operational implications are staggering. For the American healthcare landscape, where the cost of specialized labor continues to climb, adopting such a model could be the difference between a functional screening program and one that is perpetually overstretched and prone to error.
Reconceptualizing the Role of Diagnostic Technology
One of the most profound takeaways from the Swedish research is the urgent need for a conceptual shift in how the medical community and regulatory bodies categorize diagnostic technology. Historically, the FDA has viewed artificial intelligence through the narrow lens of “Clinical Decision Support,” treating it as a secondary tool designed to assist a doctor in their independent judgment. However, the MASAI results suggest that when a human and an algorithm operate as a combined unit to outperform two independent human experts, the AI has moved past being a mere assistant. It has become a core component of a superior diagnostic system. This necessitates a transition toward viewing the “human-algorithm dyad” as the actual regulated entity. Rather than evaluating software in isolation or testing the algorithm against a static dataset, the focus must shift to how the symbiotic relationship between man and machine performs in a live, clinical environment.
The current challenge lies in the fact that medical devices are still largely tested as discrete tools rather than integrated components of a broader clinical workflow. This traditional approach ignores the nuanced interactions that occur when a radiologist incorporates algorithmic feedback into their final assessment. The MASAI trial highlights that the most effective outcomes are not the result of the software acting alone, but rather the result of a specialized synergy where the algorithm compensates for human cognitive limitations, such as fatigue and pattern saturation. To reflect this reality, regulatory oversight must evolve to account for the performance of the cohesive unit. If the goal of regulation is to ensure patient safety and efficacy, then the assessment must mirror the actual conditions of use. Failing to recognize the dyad as the primary functional unit risks misinterpreting how these tools actually impact patient outcomes, potentially leading to the approval of tools that look good on paper but fail to provide meaningful benefits in practice.
Addressing the Regulatory Gap and International Divergence
FDA Inertia and the Challenges of Current Frameworks
Despite the overwhelming evidence presented by the MASAI trial, the FDA’s current regulatory architecture remains largely ill-equipped to process these systemic advancements. The agency’s existing guidelines, including the AI/ML Action Plan and various draft guidances on Predetermined Change Control Plans, focus heavily on the mechanics of software updates and prospective performance standards. While these are necessary components of oversight, they fail to address the more fundamental question of how to authorize a medical device that fundamentally alters the established standard of care. There is currently no formal, codified mechanism for the FDA to evaluate a “system” where the human and the machine are inseparable parts of the diagnostic process. This regulatory “blind spot” creates a massive structural gap between the clinical reality of 2026 and the federal oversight mechanisms that were designed for a different era of medical technology.
This inertia is not merely a matter of administrative delay; it has real-world consequences for the development and deployment of life-saving tools. Medical device sponsors often find themselves in a state of limbo, as the lack of clear pathways for human-AI integration makes the clearance process unpredictable and prohibitively expensive. When the FDA lacks a standardized framework to assess these new models, reviewers are left to make ad-hoc decisions that can vary significantly from one submission to the next. This inconsistency discourages innovation, as companies are hesitant to invest in revolutionary systems that might be rejected not because they are ineffective, but because they do not fit into the existing checkboxes. Consequently, the American public is denied access to validated technologies that have already been proven to save lives and optimize resources in other jurisdictions, effectively stalling the progress of domestic preventative medicine.
The Friction of Independent Clinical Judgment
A significant point of friction in the current regulatory landscape involves the “independent clinical judgment” anchor established by the 21st Century Cures Act. This legislation distinguishes between software that merely informs a physician’s decision and software that is intended to replace or direct that judgment. The MASAI trial results effectively blur this line by showing that the most successful clinical outcomes occur when the algorithm acts as a primary diagnostic partner rather than a passive advisor. When a radiologist relies on an algorithm to determine which scans require further review and which can be safely dismissed, the “independence” of their judgment is inextricably linked to the performance of the software. Because the FDA lacks a modern definition for this collaborative relationship, it continues to struggle with how to label and clear these devices. This creates a paradox where a tool can be proven superior in clinical trials but cannot be marketed for its true capabilities due to outdated legal definitions.
This friction is further complicated by the fact that the current FDA review process often requires developers to demonstrate that their AI does not “overly influence” the physician, a requirement that seems contradictory to the goal of improving diagnostic accuracy through technology. If the algorithm is designed to catch what the human eye misses, then its influence is precisely the source of its value. By clinging to a rigid interpretation of independent judgment, regulators are inadvertently penalizing the most effective systems. The absence of a codified standard for human-AI interaction means that the very synergy that makes the MASAI model successful is viewed as a regulatory hurdle rather than a clinical breakthrough. To resolve this, the medical community must push for a reinterpretation of clinical judgment that acknowledges the reality of modern practice, where the expertise of the doctor is augmented, not replaced, by the precision of the algorithm.
The Growing Divide Between American and European Policy
There is a widening divergence between the regulatory postures of the United States and the European Union, which has significant implications for global health equity and innovation. The European Union’s AI Act, which has already entered a phase of structured enforcement, classifies AI used in medical screening as “high-risk” but provides a much more flexible framework for its implementation. Unlike the American system, the European model incorporates “conformity assessments” that allow for the use of post-market performance data to maintain and update compliance. This proactive approach has enabled European regulators to move much faster in clearing AI-augmented mammography systems that utilize the single-reader model. As a result, international firms often possess clinical data and performance claims that far exceed what they are permitted to state on their United States labels, creating a two-tiered system of medical care.
This policy gap has led to a scenario where American patients and providers are operating with a version of technology that is artificially limited by regulatory constraints. While a company like iCAD or Lunit might have evidence demonstrating that their system can safely replace a second human reader, the FDA’s narrow labeling requirements often force them to market their products only for “detection assistance.” This discrepancy not only limits the operational benefits of the technology but also creates confusion among practitioners who see the international success of these systems but are legally barred from implementing them in the same way. The divergence suggests that the U.S. is falling behind in the global race to modernize healthcare, potentially leading to a “brain drain” of medical technology companies moving their primary operations and first-to-market launches to regions with more progressive and predictable regulatory environments.
Strategic Recommendations for Modernizing Oversight
Redefining Evidentiary Standards for System Superiority
To effectively bridge the gap between scientific advancement and federal regulation, the FDA’s Digital Health Center of Excellence must prioritize the creation of guidance documents that specifically address AI-augmented systems as singular clinical units. These documents should establish clear, minimum requirements for “system superiority” claims, providing developers with a predictable roadmap for validation. Rather than relying on traditional retrospective data, the agency should encourage the use of pre-specified reader study designs that mimic the MASAI trial’s methodology. By setting rigorous statistical thresholds for these integrated systems, the FDA can ensure that any technology cleared for use has been proven to enhance, rather than merely change, the diagnostic process. This transition would allow the agency to move away from case-by-case guesswork and toward a standardized, science-based approach that recognizes the unique nature of human-AI collaboration.
Furthermore, these new standards should include specific protocols for testing the robustness of the dyad across diverse patient populations and imaging hardware. One of the primary concerns with AI is its ability to maintain performance in different clinical settings; therefore, the evidentiary bar must include multi-site validation that accounts for real-world variability. By requiring developers to prove that the human-algorithm unit remains effective across different demographics and equipment types, the FDA can mitigate the risk of algorithmic bias and ensure equitable care. Providing this level of clarity would not only protect patients but also streamline the path to market for high-quality innovations. When developers know exactly what data is required to prove system superiority, they can focus their resources on generating that evidence, ultimately accelerating the delivery of life-saving diagnostic tools to the clinics that need them most.
Transitioning to Outcome-Based Validation Methods
A critical step in evolving the regulatory framework is shifting the focus of screening trials from “concurrent pathology” to “follow-up cancer incidence.” Traditionally, the FDA has evaluated screening tools based on what a doctor and the software can see in the moment—a metric that provides an incomplete picture of a tool’s true impact. The MASAI trial, however, utilized a longitudinal approach that tracked “interval cancers,” which are cases that were missed during the initial screening but appeared before the next scheduled exam. This outcome-based validation is the only true way to measure whether a diagnostic system is actually improving the health of a population. By adopting this standard, the FDA would align its requirements with the most rigorous scientific methods available, ensuring that approved devices are not just finding more anomalies, but are specifically finding the dangerous cancers that currently slip through the cracks of the human-only model.
Implementing this shift would require a significant change in how clinical trials are structured and funded, as longitudinal studies are inherently more time-consuming and expensive than cross-sectional ones. However, the long-term benefits to public health far outweigh these initial costs. Outcome-based validation provides a level of certainty that concurrent pathology simply cannot match, as it accounts for the natural history of the disease and the real-world performance of the screening program. To support this transition, the FDA could explore the use of real-world evidence and post-market surveillance as a way to supplement initial clearance data. This would allow for a more dynamic regulatory process where a device is granted conditional approval based on strong preliminary data, with full clearance contingent on the successful tracking of long-term patient outcomes. Such a model would ensure that the US regulatory system remains grounded in the highest levels of scientific integrity.
Responding to the Urgency of the Healthcare Crisis
The final and perhaps most urgent necessity for regulatory evolution is the implementation of time-bound responses that reflect the current reality of the healthcare workforce crisis. As radiologists retire faster than the next generation can be trained, the backlog for preventative screenings continues to grow, placing millions of patients at risk. The MASAI trial has already provided the proof of concept: healthcare systems can maintain, and even improve, the standard of care with fewer human resources, provided the technology is integrated correctly. The FDA must move beyond “thoughtful frameworks” and toward actionable clearance decisions that allow hospitals to deploy these systems without unnecessary administrative delay. Every month spent in regulatory limbo is a month where clinics remain understaffed and cancers go undetected due to overstretched resources.
In conclusion, the findings from the MASAI trial served as a catalyst for a long-overdue conversation about the future of medical regulation. The study successfully demonstrated that the integration of artificial intelligence into mammography is no longer a futuristic concept but a present-day necessity for maintaining a viable screening infrastructure. To capitalize on these advancements, the FDA must modernize its approach by defining human-algorithm dyads as regulated units, shifting toward outcome-based validation, and responding with the urgency required by the national specialist shortage. By aligning regulatory policy with the reality of 21st-century medicine, the agency can ensure that the United States remains at the forefront of clinical innovation while providing patients with the most accurate and efficient care possible. The path forward requires a bold departure from traditional silos, favoring an integrated model that reflects the true potential of the man-machine partnership.